| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2006-08-13 13:21:05 UTC |
|---|
| Update Date | 2022-03-07 02:49:20 UTC |
|---|
| HMDB ID | HMDB0004162 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Galactan |
|---|
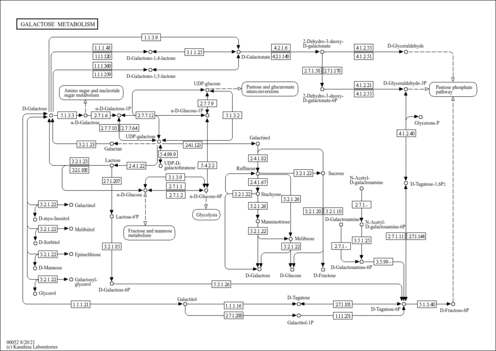
| Description | Galactan is a beta-glucan consisting of polymerized galactose. Beta -glucans are glucose polymers found in the cell walls of plants, fungi, and bacteria and as conserved structures can be considered to be classical pathogen-associated molecular patterns. These polymers belong to a class of drugs known as biological response modifiers and have a variety of effects on the immune system, including antitumor and anti-infective activities, protecting against fungal, bacterial, viral, and protozoal infections. Both particulate and soluble beta -glucans mediate these activities by activating leukocytes and stimulating their phagocytic activity and the production of reactive oxygen intermediates, inflammatory mediators, and cytokines. Despite the wealth of knowledge regarding the effects of beta-glucans and their obvious therapeutic potential, the molecular mechanisms that underlie these responses are not well understood. A number of receptors are thought to play a role in mediating these responses, including murine Dectin-1, which we recently identified as a beta-glucan receptor. Dectin-1 is a C-type lectin receptor that mediates the production of reactive oxygen species and inflammatory cytokines. Galactan is found in hemicellulose and can be converted to galactose by hydrolysis. (PMID: 11567029 , 11567029 , 16880608 ). Galactan can be produced by bacteria, such as Bifidobacteria, Lactococcus and Methylobacterium (http://www.scielo.br/pdf/babt/v54n6/02.pdf). |
|---|
| Structure | CO[C@@H]1O[C@H](CO)[C@H](O[C@@H]2O[C@H](CO)[C@H](OC)[C@H](O)[C@H]2O)[C@H](O)[C@H]1O InChI=1S/C14H26O11/c1-21-11-5(3-15)24-14(10(20)7(11)17)25-12-6(4-16)23-13(22-2)9(19)8(12)18/h5-20H,3-4H2,1-2H3/t5-,6-,7-,8-,9-,10-,11+,12+,13-,14+/m1/s1 |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | (C12H20O11)nC2H6 |
|---|
| Average Molecular Weight | Not Available |
|---|
| Monoisotopic Molecular Weight | Not Available |
|---|
| IUPAC Name | Not Available |
|---|
| Traditional Name | Not Available |
|---|
| CAS Registry Number | 39300-87-3 |
|---|
| SMILES | CO[C@@H]1O[C@H](CO)[C@H](O[C@@H]2O[C@H](CO)[C@H](OC)[C@H](O)[C@H]2O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C14H26O11/c1-21-11-5(3-15)24-14(10(20)7(11)17)25-12-6(4-16)23-13(22-2)9(19)8(12)18/h5-20H,3-4H2,1-2H3/t5-,6-,7-,8-,9-,10-,11+,12+,13-,14+/m1/s1 |
|---|
| InChI Key | PTHCMJGKKRQCBF-ICIGWGHZSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as o-glycosyl compounds. These are glycoside in which a sugar group is bonded through one carbon to another group via a O-glycosidic bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | O-glycosyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - O-glycosyl compound
- Disaccharide
- Oxane
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Ether
- Dialkyl ether
- Acetal
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross Sections| Predictor | Adduct Type | CCS Value (Å2) | Reference |
|---|
| DeepCCS | [M+H]+ | 179.332 | 30932474 | | DeepCCS | [M-H]- | 177.309 | 30932474 | | DeepCCS | [M-2H]- | 210.764 | 30932474 | | DeepCCS | [M+Na]+ | 185.205 | 30932474 |
Predicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 1.75 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 9.9223 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 7.74 minutes | 32390414 | | AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 264.2 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 1124.5 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 218.6 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 61.2 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 179.6 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 51.3 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 288.3 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 289.2 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 688.0 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 622.1 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 94.2 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 906.5 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 222.6 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 227.3 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 491.2 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 363.1 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 242.1 seconds | 40023050 |
Predicted Kovats Retention IndicesNot Available |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Galactan GC-MS (Non-derivatized) - 70eV, Positive | splash10-0k9i-3329000000-2b7bd69033e8a4fea64a | 2017-11-06 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - Galactan Quattro_QQQ 10V, Positive-QTOF (Annotated) | splash10-00di-0009000000-622a9ee87e03229ca3a1 | 2012-07-25 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Galactan Quattro_QQQ 25V, Positive-QTOF (Annotated) | splash10-00kf-2595000000-b946c2ed9bf4a89bde21 | 2012-07-25 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Galactan Quattro_QQQ 40V, Positive-QTOF (Annotated) | splash10-0ik9-2900000000-e1be501a9ca553503d2d | 2012-07-25 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 10V, Positive-QTOF | splash10-0fi1-0907000000-f53409baaccf2c283452 | 2019-02-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 20V, Positive-QTOF | splash10-004j-0901000000-c35685818fdd351d012f | 2019-02-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 40V, Positive-QTOF | splash10-004j-3900000000-5d3982d1bb24102880b8 | 2019-02-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 10V, Negative-QTOF | splash10-014l-0619000000-ee637f461d7c96fadf3e | 2019-02-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 20V, Negative-QTOF | splash10-002f-2902000000-5683d5f6382d37900c3c | 2019-02-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 40V, Negative-QTOF | splash10-01ox-6900000000-db49a499d8280d28c64f | 2019-02-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 10V, Positive-QTOF | splash10-00di-0609000000-0dcb8fef7c013777f12e | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 20V, Positive-QTOF | splash10-004j-1900000000-3ea35321e04f2cba1d3a | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 40V, Positive-QTOF | splash10-0bvj-9710000000-bb5f9bcfdec86cf6b78a | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 10V, Negative-QTOF | splash10-014i-0209000000-59d4bec3247cdead5672 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 20V, Negative-QTOF | splash10-016r-4948000000-7a4b110014b05330fed4 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Galactan 40V, Negative-QTOF | splash10-0596-9310000000-b79abc5ca8f54ee5ef9c | 2021-09-24 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum |
|
|---|
| General References | - Vierbuchen M, Bohmer G, Uhlenbruck G, Fischer R: Immuno- and histochemical studies on galactan and human blood group-related receptors in the bovine lung. Immunobiology. 1986 Aug;172(1-2):11-20. [PubMed:3095224 ]
- Uhlenbruck G, Karduck D, Haupt H, Schwick HG: C-reactive protein (CRP), 9.5 salpha1-glycoprotein and C1q: serum proteins with lectin properties? Z Immunitatsforsch Immunobiol. 1979 Feb;155(3):262-6. [PubMed:425611 ]
- Willment JA, Gordon S, Brown GD: Characterization of the human beta -glucan receptor and its alternatively spliced isoforms. J Biol Chem. 2001 Nov 23;276(47):43818-23. Epub 2001 Sep 20. [PubMed:11567029 ]
- Kato Y, Adachi Y, Ohno N: Contribution of N-linked oligosaccharides to the expression and functions of beta-glucan receptor, Dectin-1. Biol Pharm Bull. 2006 Aug;29(8):1580-6. [PubMed:16880608 ]
|
|---|