| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2023-02-21 17:15:26 UTC |
|---|
| HMDB ID | HMDB0001096 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Carbamoyl phosphate |
|---|
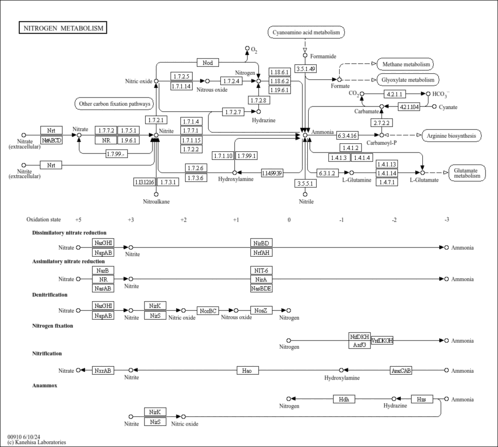
| Description | Carbamoyl phosphate, also known as carbamoyl-p or phosphate, carbamyl, belongs to the class of organic compounds known as organic phosphoric acids and derivatives. These are organic compounds containing phosphoric acid or a derivative thereof. Carbamoyl phosphate is a moderately acidic compound (based on its pKa). Carbamoyl phosphate exists in all living species, ranging from bacteria to humans. Within humans, carbamoyl phosphate participates in a number of enzymatic reactions. In particular, carbamoyl phosphate can be biosynthesized from L-glutamine and hydrogen carbonate through the action of the enzyme cad protein. In addition, carbamoyl phosphate can be converted into ureidosuccinic acid through the action of the enzyme cad protein. Its enzymatic counterpart, carbamoyl phosphate synthetase I (CPS I), interacts with a class of molecules called sirtuins, NAD dependent protein deacetylases, and ATP to form carbamoyl phosphate. It is produced from bicarbonate, ammonia (derived from amino acids), and phosphate (from ATP). In humans, carbamoyl phosphate is involved in the metabolic disorder called the beta-ureidopropionase deficiency pathway. Outside of the human body, Carbamoyl phosphate has been detected, but not quantified in, several different foods, such as cucurbita, peanuts, common oregano, figs, and chinese water chestnuts. This could make carbamoyl phosphate a potential biomarker for the consumption of these foods. Carbamoyl phosphate is a potentially toxic compound. CP then enters the urea cycle in which it reacts with ornithine (a process catalyzed by the enzyme ornithine transcarbamylase) to form citrulline. A defect in the CPS I enzyme, and a subsequent deficiency in the production of carbamoyl phosphate has been linked to hyper-ammonemia in humans. In land-dwelling animals, it is an intermediary metabolite in nitrogen disposal through the urea cycle and the synthesis of pyrimidines. Carbamoyl phosphate is an anion of biochemical significance. |
|---|
| Structure | InChI=1S/CH4NO5P/c2-1(3)7-8(4,5)6/h(H2,2,3)(H2,4,5,6) |
|---|
| Synonyms | | Value | Source |
|---|
| Aminocarbonyl dihydrogen phosphate | ChEBI | | Carbamic phosphoric monoanhydride | ChEBI | | Carbamyl phosphate | ChEBI | | Monocarbamoyl phosphate | ChEBI | | Phosphoric acid mono(formamide)ester | ChEBI | | Aminocarbonyl dihydrogen phosphoric acid | Generator | | Carbamoyl phosphoric acid | Generator | | Carbamyl phosphoric acid | Generator | | Monocarbamoyl phosphoric acid | Generator | | Phosphate mono(formamide)ester | Generator | | Carbamic acid monoanhydride with phosphorate | HMDB | | Carbamic acid monoanhydride with phosphoric acid | HMDB | | Carbamoyl-P | HMDB | | Carbamoyl-phosphate | HMDB | | Carbamoylphosphate | HMDB | | Carbamyl-phosphate | HMDB | | Phosphate, carbamyl | MeSH, HMDB | | Phosphate, carbamoyl | MeSH, HMDB | | (Carbamoyloxy)phosphonic acid | HMDB | | Carbamoyl phosphate | HMDB |
|
|---|
| Chemical Formula | CH4NO5P |
|---|
| Average Molecular Weight | 141.0199 |
|---|
| Monoisotopic Molecular Weight | 140.982708755 |
|---|
| IUPAC Name | (carbamoyloxy)phosphonic acid |
|---|
| Traditional Name | carbamoyl-phosphate |
|---|
| CAS Registry Number | 590-55-6 |
|---|
| SMILES | NC(=O)OP(O)(O)=O |
|---|
| InChI Identifier | InChI=1S/CH4NO5P/c2-1(3)7-8(4,5)6/h(H2,2,3)(H2,4,5,6) |
|---|
| InChI Key | FFQKYPRQEYGKAF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as organic phosphoric acids and derivatives. These are organic compounds containing phosphoric acid or a derivative thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Organic phosphoric acids and derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Organic phosphoric acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Organic phosphoric acid derivative
- Carboximidic acid derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Imine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 1.69 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 8.4643 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 7.76 minutes | 32390414 | | AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 436.6 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 444.5 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 355.8 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 61.8 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 263.2 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 163.9 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 262.5 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 247.1 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 904.2 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 554.4 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 40.8 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 568.1 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 217.4 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 391.9 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 834.8 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 534.7 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 476.0 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Carbamoyl phosphate,1TMS,isomer #1 | C[Si](C)(C)OP(=O)(O)OC(N)=O | 1352.9 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,1TMS,isomer #1 | C[Si](C)(C)OP(=O)(O)OC(N)=O | 1422.2 | Standard non polar | 33892256 | | Carbamoyl phosphate,1TMS,isomer #1 | C[Si](C)(C)OP(=O)(O)OC(N)=O | 2583.0 | Standard polar | 33892256 | | Carbamoyl phosphate,1TMS,isomer #2 | C[Si](C)(C)NC(=O)OP(=O)(O)O | 1473.0 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,1TMS,isomer #2 | C[Si](C)(C)NC(=O)OP(=O)(O)O | 1556.5 | Standard non polar | 33892256 | | Carbamoyl phosphate,1TMS,isomer #2 | C[Si](C)(C)NC(=O)OP(=O)(O)O | 2830.2 | Standard polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #1 | C[Si](C)(C)OP(=O)(OC(N)=O)O[Si](C)(C)C | 1414.9 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #1 | C[Si](C)(C)OP(=O)(OC(N)=O)O[Si](C)(C)C | 1515.6 | Standard non polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #1 | C[Si](C)(C)OP(=O)(OC(N)=O)O[Si](C)(C)C | 2321.0 | Standard polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #2 | C[Si](C)(C)NC(=O)OP(=O)(O)O[Si](C)(C)C | 1473.5 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #2 | C[Si](C)(C)NC(=O)OP(=O)(O)O[Si](C)(C)C | 1577.8 | Standard non polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #2 | C[Si](C)(C)NC(=O)OP(=O)(O)O[Si](C)(C)C | 2269.4 | Standard polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #3 | C[Si](C)(C)N(C(=O)OP(=O)(O)O)[Si](C)(C)C | 1570.8 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #3 | C[Si](C)(C)N(C(=O)OP(=O)(O)O)[Si](C)(C)C | 1682.9 | Standard non polar | 33892256 | | Carbamoyl phosphate,2TMS,isomer #3 | C[Si](C)(C)N(C(=O)OP(=O)(O)O)[Si](C)(C)C | 2323.9 | Standard polar | 33892256 | | Carbamoyl phosphate,3TMS,isomer #1 | C[Si](C)(C)NC(=O)OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C | 1539.9 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,3TMS,isomer #1 | C[Si](C)(C)NC(=O)OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C | 1628.6 | Standard non polar | 33892256 | | Carbamoyl phosphate,3TMS,isomer #1 | C[Si](C)(C)NC(=O)OP(=O)(O[Si](C)(C)C)O[Si](C)(C)C | 1978.8 | Standard polar | 33892256 | | Carbamoyl phosphate,3TMS,isomer #2 | C[Si](C)(C)OP(=O)(O)OC(=O)N([Si](C)(C)C)[Si](C)(C)C | 1575.5 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,3TMS,isomer #2 | C[Si](C)(C)OP(=O)(O)OC(=O)N([Si](C)(C)C)[Si](C)(C)C | 1628.3 | Standard non polar | 33892256 | | Carbamoyl phosphate,3TMS,isomer #2 | C[Si](C)(C)OP(=O)(O)OC(=O)N([Si](C)(C)C)[Si](C)(C)C | 1875.0 | Standard polar | 33892256 | | Carbamoyl phosphate,4TMS,isomer #1 | C[Si](C)(C)OP(=O)(OC(=O)N([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1605.8 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,4TMS,isomer #1 | C[Si](C)(C)OP(=O)(OC(=O)N([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1702.6 | Standard non polar | 33892256 | | Carbamoyl phosphate,4TMS,isomer #1 | C[Si](C)(C)OP(=O)(OC(=O)N([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1702.6 | Standard polar | 33892256 | | Carbamoyl phosphate,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC(N)=O | 1638.2 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC(N)=O | 1650.0 | Standard non polar | 33892256 | | Carbamoyl phosphate,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC(N)=O | 2824.5 | Standard polar | 33892256 | | Carbamoyl phosphate,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O)O | 1731.1 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O)O | 1714.8 | Standard non polar | 33892256 | | Carbamoyl phosphate,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O)O | 2968.8 | Standard polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(OC(N)=O)O[Si](C)(C)C(C)(C)C | 1838.0 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(OC(N)=O)O[Si](C)(C)C(C)(C)C | 1918.9 | Standard non polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(OC(N)=O)O[Si](C)(C)C(C)(C)C | 2674.8 | Standard polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O)O[Si](C)(C)C(C)(C)C | 1915.7 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O)O[Si](C)(C)C(C)(C)C | 1929.6 | Standard non polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O)O[Si](C)(C)C(C)(C)C | 2587.6 | Standard polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N(C(=O)OP(=O)(O)O)[Si](C)(C)C(C)(C)C | 1984.5 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N(C(=O)OP(=O)(O)O)[Si](C)(C)C(C)(C)C | 2030.0 | Standard non polar | 33892256 | | Carbamoyl phosphate,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)N(C(=O)OP(=O)(O)O)[Si](C)(C)C(C)(C)C | 2416.1 | Standard polar | 33892256 | | Carbamoyl phosphate,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2138.2 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2121.9 | Standard non polar | 33892256 | | Carbamoyl phosphate,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC(=O)OP(=O)(O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2417.9 | Standard polar | 33892256 | | Carbamoyl phosphate,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2175.4 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2208.4 | Standard non polar | 33892256 | | Carbamoyl phosphate,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OP(=O)(O)OC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2205.0 | Standard polar | 33892256 | | Carbamoyl phosphate,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(OC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2360.9 | Semi standard non polar | 33892256 | | Carbamoyl phosphate,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(OC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2392.6 | Standard non polar | 33892256 | | Carbamoyl phosphate,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OP(=O)(OC(=O)N([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2148.2 | Standard polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Carbamoyl phosphate GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-9100000000-6a951539300f7f5b64d6 | 2016-09-22 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Carbamoyl phosphate GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Carbamoyl phosphate GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 10V, Positive-QTOF | splash10-0002-9000000000-1e9b8619eed467aca3cc | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 20V, Positive-QTOF | splash10-0002-9000000000-49d39aaea7b7b239f046 | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 40V, Positive-QTOF | splash10-0005-9000000000-ff8b83b8641d1d23539f | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 10V, Negative-QTOF | splash10-002b-9100000000-9b7601209f4777504c50 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 20V, Negative-QTOF | splash10-004i-9000000000-068148ee9b7b7acd6692 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 40V, Negative-QTOF | splash10-004i-9000000000-2eef12354837d65af0c1 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 10V, Positive-QTOF | splash10-0002-9200000000-df9df6c92477a4e65654 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 20V, Positive-QTOF | splash10-0002-9000000000-82163424a165525ca974 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 40V, Positive-QTOF | splash10-0006-9000000000-959c63a896bb7c629164 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 10V, Negative-QTOF | splash10-004i-9000000000-a5e502a2627af2048a1f | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 20V, Negative-QTOF | splash10-004i-9000000000-a5e502a2627af2048a1f | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Carbamoyl phosphate 40V, Negative-QTOF | splash10-004i-9000000000-a5e502a2627af2048a1f | 2021-09-24 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum |
IR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M-H]-) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+H]+) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+Na]+) | 2023-02-03 | FELIX lab | View Spectrum |
|
|---|
| General References | - Sigoillot FD, Kotsis DH, Serre V, Sigoillot SM, Evans DR, Guy HI: Nuclear localization and mitogen-activated protein kinase phosphorylation of the multifunctional protein CAD. J Biol Chem. 2005 Jul 8;280(27):25611-20. Epub 2005 May 12. [PubMed:15890648 ]
- Struck J, Uhlein M, Morgenthaler NG, Furst W, Hoflich C, Bahrami S, Bergmann A, Volk HD, Redl H: Release of the mitochondrial enzyme carbamoyl phosphate synthase under septic conditions. Shock. 2005 Jun;23(6):533-8. [PubMed:15897806 ]
- Schnater JM, Bruder E, Bertschin S, Woodtli T, de Theije C, Pietsch T, Aronson DC, von Schweinitz D, Lamers WH, Kohler ES: Subcutaneous and intrahepatic growth of human hepatoblastoma in immunodeficient mice. J Hepatol. 2006 Sep;45(3):377-86. Epub 2006 May 3. [PubMed:16780998 ]
- Chen KF, Lai YY, Sun HS, Tsai SJ: Transcriptional repression of human cad gene by hypoxia inducible factor-1alpha. Nucleic Acids Res. 2005 Sep 9;33(16):5190-8. Print 2005. [PubMed:16155188 ]
|
|---|