| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2023-02-21 17:14:27 UTC |
|---|
| HMDB ID | HMDB0000005 |
|---|
| Secondary Accession Numbers | - HMDB00005
- HMDB0006544
- HMDB06544
|
|---|
| Metabolite Identification |
|---|
| Common Name | 2-Ketobutyric acid |
|---|
| Description | 2-Ketobutyric acid, also known as alpha-ketobutyrate or 2-oxobutyrate, belongs to the class of organic compounds known as short-chain keto acids and derivatives. These are keto acids with an alkyl chain the contains less than 6 carbon atoms. 2-Ketobutyric acid is a substance that is involved in the metabolism of many amino acids (glycine, methionine, valine, leucine, serine, threonine, isoleucine) as well as propanoate metabolism and C-5 branched dibasic acid metabolism. It is also one of the degradation products of threonine. It can be converted into propionyl-CoA (and subsequently methylmalonyl CoA, which can be converted into succinyl CoA, a citric acid cycle intermediate), and thus enter the citric acid cycle. More specifically, 2-ketobutyric acid is a product of the lysis of cystathionine. |
|---|
| Structure | InChI=1S/C4H6O3/c1-2-3(5)4(6)7/h2H2,1H3,(H,6,7) |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Ketobutanoic acid | ChEBI | | 2-Oxobutyric acid | ChEBI | | 3-Methyl pyruvic acid | ChEBI | | alpha-Ketobutyrate | ChEBI | | alpha-Ketobutyric acid | ChEBI | | alpha-oxo-N-Butyric acid | ChEBI | | 2-Oxobutyrate | Kegg | | 2-Oxobutanoic acid | Kegg | | 2-Ketobutanoate | Generator | | 3-Methyl pyruvate | Generator | | a-Ketobutyrate | Generator | | a-Ketobutyric acid | Generator | | Α-ketobutyrate | Generator | | Α-ketobutyric acid | Generator | | a-oxo-N-Butyrate | Generator | | a-oxo-N-Butyric acid | Generator | | alpha-oxo-N-Butyrate | Generator | | Α-oxo-N-butyrate | Generator | | Α-oxo-N-butyric acid | Generator | | 2-Oxobutanoate | Generator | | 2-Ketobutyrate | Generator | | 2-oxo-Butanoate | HMDB | | 2-oxo-Butanoic acid | HMDB | | 2-oxo-Butyrate | HMDB | | 2-oxo-Butyric acid | HMDB | | 2-oxo-N-Butyrate | HMDB | | 2-oxo-N-Butyric acid | HMDB | | 3-Methylpyruvate | HMDB | | 3-Methylpyruvic acid | HMDB | | a-Keto-N-butyrate | HMDB | | a-Keto-N-butyric acid | HMDB | | a-Oxobutyrate | HMDB | | a-Oxobutyric acid | HMDB | | alpha-Keto-N-butyrate | HMDB | | alpha-Keto-N-butyric acid | HMDB | | alpha-Ketobutric acid | HMDB | | alpha-Oxobutyrate | HMDB | | alpha-Oxobutyric acid | HMDB | | Methyl-pyruvate | HMDB | | Methyl-pyruvic acid | HMDB | | Propionyl-formate | HMDB | | Propionyl-formic acid | HMDB | | alpha-Ketobutyric acid, sodium salt | HMDB | | 2-Ketobutyric acid | ChEBI |
|
|---|
| Chemical Formula | C4H6O3 |
|---|
| Average Molecular Weight | 102.0886 |
|---|
| Monoisotopic Molecular Weight | 102.031694058 |
|---|
| IUPAC Name | 2-oxobutanoic acid |
|---|
| Traditional Name | 2-oxobutanoic acid |
|---|
| CAS Registry Number | 600-18-0 |
|---|
| SMILES | CCC(=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C4H6O3/c1-2-3(5)4(6)7/h2H2,1H3,(H,6,7) |
|---|
| InChI Key | TYEYBOSBBBHJIV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as short-chain keto acids and derivatives. These are keto acids with an alkyl chain the contains less than 6 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Keto acids and derivatives |
|---|
| Sub Class | Short-chain keto acids and derivatives |
|---|
| Direct Parent | Short-chain keto acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Short-chain keto acid
- Alpha-keto acid
- Alpha-hydroxy ketone
- Ketone
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 33 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 119 mg/mL | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 2.14 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 10.1413 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 2.94 minutes | 32390414 | | AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 102.8 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 1229.1 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 375.4 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 112.4 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 252.8 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 80.3 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 329.7 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 392.6 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 136.9 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 740.0 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 268.5 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 961.3 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 247.8 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 300.6 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 572.0 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 295.1 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 189.7 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 2-Ketobutyric acid,1TMS,isomer #1 | CCC(=O)C(=O)O[Si](C)(C)C | 1013.7 | Semi standard non polar | 33892256 | | 2-Ketobutyric acid,1TMS,isomer #2 | CC=C(O[Si](C)(C)C)C(=O)O | 1091.1 | Semi standard non polar | 33892256 | | 2-Ketobutyric acid,2TMS,isomer #1 | CC=C(O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1182.5 | Semi standard non polar | 33892256 | | 2-Ketobutyric acid,2TMS,isomer #1 | CC=C(O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1154.8 | Standard non polar | 33892256 | | 2-Ketobutyric acid,2TMS,isomer #1 | CC=C(O[Si](C)(C)C)C(=O)O[Si](C)(C)C | 1200.1 | Standard polar | 33892256 | | 2-Ketobutyric acid,1TBDMS,isomer #1 | CCC(=O)C(=O)O[Si](C)(C)C(C)(C)C | 1240.5 | Semi standard non polar | 33892256 | | 2-Ketobutyric acid,1TBDMS,isomer #2 | CC=C(O[Si](C)(C)C(C)(C)C)C(=O)O | 1332.4 | Semi standard non polar | 33892256 | | 2-Ketobutyric acid,2TBDMS,isomer #1 | CC=C(O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1617.2 | Semi standard non polar | 33892256 | | 2-Ketobutyric acid,2TBDMS,isomer #1 | CC=C(O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1574.5 | Standard non polar | 33892256 | | 2-Ketobutyric acid,2TBDMS,isomer #1 | CC=C(O[Si](C)(C)C(C)(C)C)C(=O)O[Si](C)(C)C(C)(C)C | 1537.5 | Standard polar | 33892256 |
|
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-0gbj-1900000000-8daa299f5bc38a7f9b3e | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-014s-1910000000-d01bcd7b41fdcc634eea | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-MS (1 MEOX; 1 TMS) | splash10-000i-9200000000-3b3816ffee5771215904 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-MS (1 MEOX; 1 TMS) | splash10-000i-6910000000-5b264d5f7e53f3e80b12 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-MS (2 TMS) | splash10-001i-6950000000-db7f6a0f03a8cce59bae | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid EI-B (Non-derivatized) | splash10-0ftb-0920000000-a6c93d8761ef15f7f7d4 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid EI-B (Non-derivatized) | splash10-014i-0920000000-406fe70e72dbd9c3f274 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-EI-TOF (Non-derivatized) | splash10-0gbj-1900000000-8daa299f5bc38a7f9b3e | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-EI-TOF (Non-derivatized) | splash10-014s-1910000000-d01bcd7b41fdcc634eea | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-MS (Non-derivatized) | splash10-000i-9200000000-3b3816ffee5771215904 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-MS (Non-derivatized) | splash10-000i-6910000000-5b264d5f7e53f3e80b12 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-MS (Non-derivatized) | splash10-001i-6950000000-db7f6a0f03a8cce59bae | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-EI-TOF (Non-derivatized) | splash10-000i-8900000000-97bab6630de4df095a7f | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 2-Ketobutyric acid GC-EI-TOF (Non-derivatized) | splash10-000i-9200000000-0ebb2354fe3f935fe177 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 2-Ketobutyric acid GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a6r-9000000000-32269ef3ef778c482f99 | 2016-09-22 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 2-Ketobutyric acid GC-MS (1 TMS) - 70eV, Positive | splash10-0adi-9200000000-f92941e9f326d8ccf3e4 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 2-Ketobutyric acid GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 2-Ketobutyric acid GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 2-Ketobutyric acid GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 2-Ketobutyric acid GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | 2021-11-05 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid Quattro_QQQ 10V, Negative-QTOF (Annotated) | splash10-0pb9-9600000000-53ebb7164f74d7d92a4d | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid Quattro_QQQ 25V, Negative-QTOF (Annotated) | splash10-0zfr-5900000000-1db864754705beb79b55 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid Quattro_QQQ 40V, Negative-QTOF (Annotated) | splash10-0udi-9800000000-5ba5b9ede5b1bc389b87 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Negative-QTOF | splash10-0udi-0900000000-bfdcad5bc3ed586bdede | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid LC-ESI-QTOF , negative-QTOF | splash10-0udi-0900000000-bfdcad5bc3ed586bdede | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid QqQ 4V, positive-QTOF | splash10-0pb9-9600000000-b10635fbec5572d4d084 | 2020-07-22 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid QqQ 8V, positive-QTOF | splash10-0a4i-9100000000-4c5e7d24dca6677e1570 | 2020-07-22 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid QqQ 12V, positive-QTOF | splash10-0a4i-9000000000-ded44cfcbe7960954fdf | 2020-07-22 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid QqQ 16V, positive-QTOF | splash10-0a4i-9000000000-1b350be7438b08f1946a | 2020-07-22 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 2-Ketobutyric acid QqQ 20V, positive-QTOF | splash10-056r-9000000000-172a175b8b9ebfd5529f | 2020-07-22 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 10V, Positive-QTOF | splash10-0k9i-9400000000-0061f4635858d851f127 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 20V, Positive-QTOF | splash10-0a4r-9100000000-b943445194fb5f8e3a0f | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 40V, Positive-QTOF | splash10-0a4i-9000000000-bc5920b9bd140be6b06d | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 10V, Negative-QTOF | splash10-0udi-5900000000-f7dfb4554f0b9e1499c9 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 20V, Negative-QTOF | splash10-0a59-9200000000-e54106ad5bf45a9f072c | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 40V, Negative-QTOF | splash10-0a4i-9000000000-7bbba57578cf720974e3 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 10V, Positive-QTOF | splash10-0a4i-9000000000-ca1ed9c5aadcbdabc2a3 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 20V, Positive-QTOF | splash10-0a4i-9000000000-aced8e004436190f07b7 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 40V, Positive-QTOF | splash10-0a4i-9000000000-a3d7ddf227bd6aa91a54 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 10V, Negative-QTOF | splash10-0pb9-9700000000-503296025148ec314383 | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 20V, Negative-QTOF | splash10-0a4i-9000000000-933b567955b5de5b1744 | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 2-Ketobutyric acid 40V, Negative-QTOF | splash10-0006-9000000000-7d9db52021fefc87a855 | 2021-09-25 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Experimental 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | 2012-12-04 | Wishart Lab | View Spectrum | | Experimental 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | 2012-12-04 | Wishart Lab | View Spectrum |
IR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M-H]-) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+H]+) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+Na]+) | 2023-02-03 | FELIX lab | View Spectrum |
|
|---|
| Biological Properties |
|---|
| Cellular Locations | |
|---|
| Biospecimen Locations | - Blood
- Cerebrospinal Fluid (CSF)
- Saliva
- Urine
|
|---|
| Tissue Locations | Not Available |
|---|
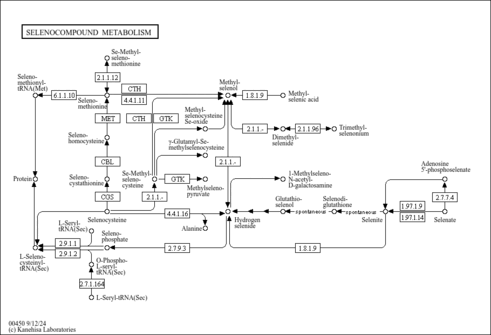
| Pathways | |
|---|
| Normal Concentrations |
|---|
| |
| Blood | Detected and Quantified | 3.33 +/- 5.00 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 9.4 (3.4-15.4) uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 8.6 (6.0-11.2) uM | Children (1-13 years old) | Both | Normal | | details | | Blood | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Cerebrospinal Fluid (CSF) | Detected and Quantified | 4.8 (3.4-6.2) uM | Children (1-13 years old) | Both | Normal | | details | | Saliva | Detected and Quantified | 0.0412 +/- 0.214 uM | Adult (>18 years old) | Male | Normal | | details | | Saliva | Detected and Quantified | 2.62 +/- 4.13 uM | Adult (>18 years old) | Not Specified | Normal | | details | | Saliva | Detected and Quantified | 4.06 +/- 8.39 uM | Adult (>18 years old) | Female | Normal | | details | | Saliva | Detected and Quantified | 4.12 +/- 4.41 uM | Adult (>18 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 0.39 (0.05-2.2) umol/mmol creatinine | Adult (>18 years old) | Male | Normal | | details | | Urine | Detected and Quantified | 0.13 (0.04-0.51) umol/mmol creatinine | Adult (>18 years old) | Female | Normal | | details | | Urine | Detected and Quantified | 2.6 (1.1-4.1) umol/mmol creatinine | Adult (>18 years old) | Male | Normal | | details | | Urine | Detected and Quantified | 2.9 (0.7-5.4) umol/mmol creatinine | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details | | Urine | Detected and Quantified | 9.476 +/- 6.626 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Normal | | details | | Urine | Detected and Quantified | 0-50 umol/mmol creatinine | Newborn (0-30 days old) | Both | Normal | | details | | Urine | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details |
|
|---|
| Abnormal Concentrations |
|---|
| |
| Urine | Detected and Quantified | 6.564 +/- 7.802 umol/mmol creatinine | Children (1 - 13 years old) | Not Specified | Eosinophilic esophagitis | | details |
|
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | | Eosinophilic esophagitis |
|---|
- Slae, M., Huynh, H., Wishart, D.S. (2014). Analysis of 30 normal pediatric urine samples via NMR spectroscopy (unpublished work). NA.
|
|
|---|
| Associated OMIM IDs | - 610247 (Eosinophilic esophagitis)
|
|---|
| External Links |
|---|
| DrugBank ID | DB04553 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB030356 |
|---|
| KNApSAcK ID | C00019675 |
|---|
| Chemspider ID | 57 |
|---|
| KEGG Compound ID | C00109 |
|---|
| BioCyc ID | 2-OXOBUTANOATE |
|---|
| BiGG ID | 33889 |
|---|
| Wikipedia Link | Alpha-Ketobutyric_acid |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 58 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 30831 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | 2OBUT |
|---|
| MarkerDB ID | Not Available |
|---|
| Good Scents ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Figge, Rainer; Lux, Fabien; Raynaud, Celine; Soucaille, Philippe. Production of a-ketobutyrate by engineered Escherichia coli.PCT Int. Appl. (2006), 31pp. |
|---|
| Material Safety Data Sheet (MSDS) | Not Available |
|---|
| General References | - Sprague CL, Elfarra AA: Detection of carboxylic acids and inhibition of hippuric acid formation in rats treated with 3-butene-1,2-diol, a major metabolite of 1,3-butadiene. Drug Metab Dispos. 2003 Aug;31(8):986-92. [PubMed:12867486 ]
- Yaegaki K, Sanada K: Biochemical and clinical factors influencing oral malodor in periodontal patients. J Periodontol. 1992 Sep;63(9):783-9. [PubMed:1474480 ]
- Yaegaki K, Sanada K: Effects of a two-phase oil-water mouthwash on halitosis. Clin Prev Dent. 1992 Jan-Feb;14(1):5-9. [PubMed:1499240 ]
- Yang W, Roth KS: Defect in alpha-ketobutyrate metabolism: a new inborn error. Clin Chim Acta. 1985 Jan 30;145(2):173-82. [PubMed:3918815 ]
|
|---|